Stem cells are primal cells found in all multi-cellular organisms that retain the ability to renew themselves through mitotic cell division and can differentiate into a diverse range of specialized cell types. Research in the human stem cell field grew out of findings by Canadian scientists Ernest A. McCulloch and James E. Till in the 1960s.
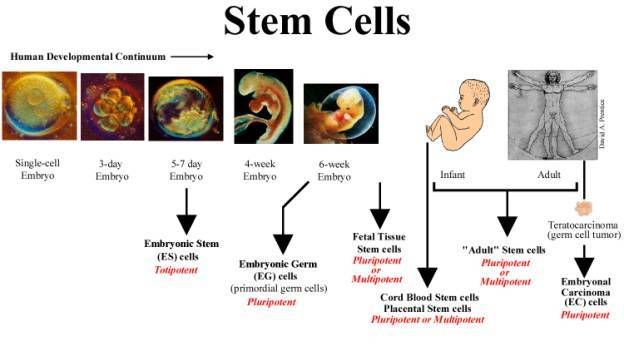
The three broad categories of mammalian stem cells are: embryonic stem cells, derived from blastocysts, adult stem cells, which are found in adult tissues, and cord blood stem cells, which are found in the umbilical cord. In a developing embryo, stem cells can differentiate into all of the specialized embryonic tissues. In adult organisms, stem cells and progenitor cells act as a repair system for the body, replenishing specialized cells.
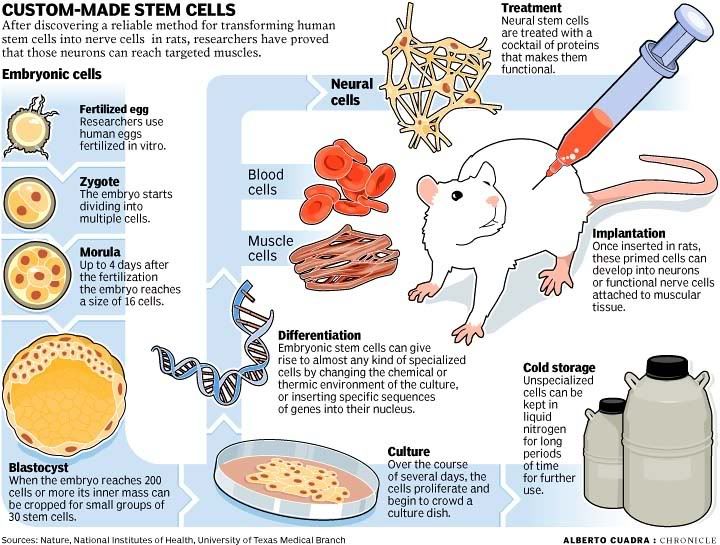
As stem cells can be readily grown and transformed into specialised cells with characteristics consistent with cells of various tissues such as muscles or nerves through cell culture, their use in medical therapies has been proposed. In particular, embryonic cell lines, autologous embryonic stem cells generated through therapeutic cloning, and highly plastic adult stem cells from the umbilical cord blood or bone marrow are touted as promising candidates
http://en.wikipedia.org/wiki/Stem_cell
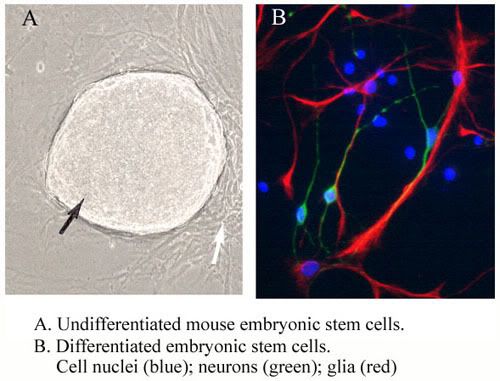
In living organisms, an undifferentiated cell that can produce other cells that eventually make up specialized tissues and organs. There are two major types of stem cells, embryonic and adult. Embryonic stem cells are located in the inner mass of a blastocyst (an embryo at a very early stage of development), and they eventually give rise to every cell type of the adult organism. Adult stem cells are found in some tissues in the adult body, such as the epidermis of the skin, the lining of the small intestine, and the bone marrow, where they serve in the regeneration of old or worn tissue. In cancer treatment, blood-forming adult stem cells are routinely harvested from bone marrow, stored, and then reinfused into patients to replace blood cells destroyed by chemotherapy or radiation therapy. This potential for replacing damaged tissues has aroused great interest in using embryonic stem cells to treat a number of other conditions, such as Parkinson disease, severe burns, and damage to the spinal cord. Mouse embryonic stem cells are widely used to create genetically modified mice that serve as models for investigating human disease. However, the use of human embryonic stem cells, which requires destroying the blastocysts from which they are obtained, has raised objections by those who feel blastocyst-stage embryos are human beings. The first human stem cell line was created in 1998, using cells harvested from embryos produced through in vitro fertilization. The use of human embryonic stem cells is allowed in some countries and prohibited or restricted in others.
http://www.answers.com/topic/stem-cell
Stem Cell Basics
http://stemcells.nih.gov/info/basics/
Media Myths
Stem Cell Research and Cloning
http://www.stemcellresearchfacts.com/media_myths.html
As widespread interest in stem cell research increases, so do the number of news articles and reports. It is an unfortunate fact that several misconceptions and distortions have taken hold within the secular media. Ten such “Media Myths” are addressed below.
Myth 1. Stem cells can only come from embryos. In fact stem cells can be taken from umbilical cords, the placenta, amniotic fluid, adult tissues and organs such as bone marrow, fat from liposuction, regions of the nose, and even from cadavers up to 20 hours after death.
Myth 2. Christians are against stem cell research. There are four categories of stem cells: embryonic stem cells, embryonic germ cells, umbilical cord stem cells, and adult stem cells. Given that germ cells can come from miscarriages that involve no deliberate interruption of pregnancy, Christians in general oppose the use of only one of these four categories, i.e., embryonic stem cells. In other words, most Christians approve of three of the four possible types of stem cell research.
Myth 3. Embryonic stem cell research has the greatest promise. Up to now, no human being has ever been cured of a disease using embryonic stem cells. Adult stem cells, on the other hand, have already cured thousands. For example, bone marrow cells from the hipbone have repaired scar tissue on the heart after heart attacks. Research using adult cells is 20-30 years ahead of embryonic stem cells and holds greater promise. This is in part because stem cells are part of the natural repair mechanisms of an adult body, while embryonic stem cells do not belong in an adult body (where they are likely to form tumors, and to be rejected as foreign tissue by the recipient). Rather, embryonic stem cells really belong only within in the specialized microenvironment of a rapidly growing embryo, which is a radically different setting from an adult body.
Myth 4. Embryonic stem cell research is against the law. In reality, there is no law or regulation against destroying human embryos for research purposes. While President Bush has banned the use of federal funding to support research on embryonic stem cell lines created after August 2001, it is not illegal. Anyone using private funds is free to pursue it.
Myth 5. President Bush created new restrictions to federal funding of embryonic stem cell research. The 1996 Dickey Amendment prohibited the use of federal funds for research that would involve the destruction of human embryos. Bush’s decision to permit research on embryonic stem cell lines created before a certain date thus relaxes this restriction from the Clinton era.
Myth 6. Theraputic cloning and reproductive cloning are fundamentally different from each other. The creation of cloned embryos either to make a baby or to harvest cells occurs by the same series of technical steps. The only difference is what will be done with the cloned human embryo that is produced. Will it be given the protection of a woman’s womb in order to be born? Or will it be destroyed for its stem cells?
Myth 7. Somatic nuclear cell transfer is different from cloning. In fact, “somatic cell nuclear transfer” is simply cloning by a different name. The end result is still a cloned embryo.
Myth 8. By doing somatic cell nuclear transfer, we can directly produce tissues or organs without having to clone an embryo. At the present stage of research, scientists are unable to bypass the creation of an embryo in the production of tissues or organs. In the future it may be possible to inject elements from the cytoplasm of a woman’s ovum into a somatic cell to “reprogram” it into a stem cell. This is called “de-differentiation.” If so, there would be no fundamental moral objection to this approach to getting stem cells.
Myth 9. Every body cell, or somatic cell, is somehow an embryo and thus a human life. People sometimes argue: “Every cell in the body has the potential to become an embryo. Does that mean that every time we wash our hands and are shedding thousands of cells, we are killing life?” The problem is that this overlooks the basic biological difference between a regular body cell, and one whose nuclear material has been fused with an unfertilized egg cell, resulting in an embryo. A normal skin cell will only give rise to more skin cells when it divides, while an embryo will give rise to the entire adult organism. Skin cells are not potential adults. Skin cells are potentially only more skin cells. Only embryos are potential adults.
Myth 10. Because frozen embryos may one day end up being discarded by somebody, that makes it allowable, even laudable, to violate and destroy those embryos. The moral analysis of what we may permissibly do with an embryo doesn’t depend on its otherwise “going to waste,” nor on the incidental fact that those embryos are “trapped” in liquid nitrogen. Consider a radical case in which a group of children are permanently trapped in a schoolhouse through no fault of their own; that would not make it morally acceptable to send in a remote control robotic device which would harvest organs from those children and cause their demise.
Stem Cell Breakthrough?
Scientists Say They Have Produced The Equivalent Of Embryonic Stem Cells In Mice.

http://www.cbsnews.com/stories/2007/06/06/tech/main2893364.shtml?source=RSSattr=HOME_2893364
Three teams of scientists say they have produced the equivalent of embryonic stem cells, at least in mice, without taking the controversial step of destroying embryos.
Their procedure makes ordinary skin cells behave like stem cells. If the same can be done with human cells — a big if — the procedure could lead to breakthrough medical treatments without the contentious ethical and political debates surrounding the use of embryos.
Embryonic stem cells can give rise to all types of tissue, so experts believe they might be used to create transplant therapies for people who are paralyzed or have illnesses ranging from diabetes to Parkinson's disease.
To harvest human embryonic stem cells, human embryos have to be destroyed, an action opposed by many people. The new studies are the latest to attempt to avoid embryo destruction.
Scientists have long hoped to find a way to reprogram ordinary body cells to act like stem cells, avoiding the use of embryos altogether. The new mouse studies seem to have accomplished that.
"I think it's one of the most exciting things that has come out about embryonic stem cells, period," said stem cell researcher Dr. Asa Abeliovich of Columbia University in New York, who didn't participate in the work. "It's very convincing that it's real."
But he and others cautioned that it will take further study to see whether this scientific advance can be harnessed for new human therapies.
"We have a long way to go,” said John Gearhart of Johns Hopkins University, a stem cell researcher who also was not involved in the new work.
In any case, it is crucial that scientists continue research with standard embryonic stem cells, said researcher Konrad Hochedlinger of the Harvard Stem Cell Institute, who led one of the three teams.
He and his colleagues present their work in the inaugural issue of the journal Cell Stem Cell. The first word in the journal's name refers to its publisher, Cell Press.
The other two teams reported their results Wednesday on the Web site of the journal Nature. Rudolf Jaenisch of the Whitehead Institute in Cambridge, Mass., is the senior author of one paper; the work behind the other paper was led by Shinya Yamanaka of Kyoto University in Japan.
The new work builds on a landmark paper Yamanaka published last August. He found that by slipping four genes into mouse skin cells called fibroblasts, he could make the altered cells behave much like embryonic stem cells in lab tests.
But these so-called "iPS" cells still showed significant differences from embryonic stem cells. The three new papers report on creating iPS cells that proved virtually identical to stem cells in a variety of lab tests.
The four inserted genes regulate the activity of other genes, which is why they can dramatically affect the cell's behavior.
Scientists caution that the experimental procedure followed in the studies would not be suitable for use in treating disease, and that it's not yet clear whether a modified version would work with human cells.
One of the inserted genes is known to promote cancer, and Yamanaka reports that mice carrying descendants of iPS cells showed tumors as a result. He and other researchers said a new approach will be needed that avoids a cancer hazard.
Gearhart called that a major issue to be resolved. In addition, he said, scientists still must show that these cells can give rise to many cell types in the lab, as embryonic stem cells can.
And all this must be accomplished in human cells — a difficult task, he said, because introducing genes into human cells is a major challenge.
If the technique can be harnessed for people, the iPS cells and the tissue they develop into would provide a genetic match to the person who donated the skin cells. That would make them suitable for transplant to that person, theoretically without fear of rejection.
Stem Cells
Stem Cell Breakthrough
http://www.eontarionow.com/science/2007/06/11/stem-cell-breakthrough/
Toronto -In what is considered a major huge breakthrough in stem cell development, researchers have found a way to make stem cells without an embryo.
Papers which were published on Tuesday show that it is possible to make stem cells out of skin cells. This would allow researchers to get around bans on using human embryonic stem cells.
This will allow new areas of research into human embryonic stem cells. Scientists know so little about human embryonic stem cells at this point that anything is possible.
Although the testing on mice was promising, researchers are still if they can do it in humans yet.
This news comes at an interesting time, just as a bill to provide federal funds for embryonic stem cell research makes its way to the president for his signature. U.S. President George Bush has made it clear that he will veto the bill, as he did similar legislation a few years ago.
Nonetheless, this new research suggests — as opponents of experimental use of embryonic stem cells have claimed all along — that there are viable alternatives to destroying human embryos in order to save lives.
Stem Cells-Rats
New Guidelines On The Way For Stem Cell Research
http://www.americanchronicle.com/articles/viewArticle.asp?articleID=29229
The stem cell controversy has ruffled the Congress, State Legislatures, President Bush, and the whole of United Nations. It has resulted in the proposal of new guidelines for stem cell research by the National Academy of Sciences, which is a non-profit group and an advisor to the government.
The guidelines demand the formation of new oversight committees at all institutions carrying out such research. These committees would comprise of researchers, law and ethics experts, and general public members.
The guidelines talk about standards for storage and distribution of cells, but do not impose a ban. There are many religious groups against the use of human embryos for research purposes. The guidelines limit the use of embryos up to the first 14 days of their growth, that is, before the cells start to differentiate into body parts.
Parameters
The supposed Embryonic Stem Cell Research Oversight Committees (ESCROs) would supervise the early embryo donations for research purpose. The guidelines establish the following parameters:
Donors will have to provide their consent and be clear in their minds that their identities would be known and they would not get any financial benefits obtained from generating stem cells.
Donors should never be lured by money in order to donate embryos.
Researchers should not force fertility clinics to develop more number of embryos than that required for reproductive treatments.
Considering the Chimeras Too
The federal government has restricted stem cell research that utilizes federal funds for cell lines developed before 2001. The guidelines of ESCRO would regulate those cell lines as well as others developed throughout the country by utilizing private or state funds.
The guidelines also take in hand chimeras. They are animals that have been infused with human cells for research. The researchers have to get the approval of ESCRO for creating chimeras, as per the guidelines. Besides this, it restricts the infusion of animal embryonic stem cells into a human embryo and vice versa.
The guidelines allow the scientists to create Chimeras ONLY when there is no other research method available. The introduction of human cells into brains of an animal would be allowed only after providing a concrete scientific justification. For example, creating mice by the infusion of human bone marrow might be allowed because it is to study blood diseases, and is important before clinical trials on humans can be done.
Overall, the ESCRO guidelines are a sort of “compromise”, after identifying the need for going beyond the usual and, at the same time, warning against the danger of going too much against the laws of nature.
Will these guidelines douse the controversial fire on stem cell research? Well, only time will tell!
The stem cell cell controversy has led to several new guidelines to ensure standards are maintained during stem cell research and public sentiments are not hurt! This is very important in the wake of new developments in cord blood research and to avoid controversies related to cord blood donors. For more details about the benefits of using a cords blood bank, process of cord blood storage and all the other information you need to know about cord blood stem cells, Visist Cord Blood Banking.
TIME.com - Stem Cells
http://www.cnn.com/SPECIALS/2001/stemcell/
Stem cell controversy
There is widespread controversy over stem cell research largely due to techniques used in the creation and usage of human embryonic stem cells. Human embryonic stem cell research is particularly controversial because, with the present state of biotechnology, most techniques used to create embryonic stem cell lines require the destruction of human embryos and/or therapeutic cloning. Some opponents of the research also argue that this practice is a slippery slope to reproductive cloning and fundamentally devalues the worth of a potential human being. Contrarily, medical researchers in the field argue that it is necessary to pursue embryonic stem cell research because the resultant technologies are expected to have significant medical potential, and that the embryos used for research are only those slated for destruction anyway. The ensuing debate has prompted authorities around the world to seek regulatory frameworks and highlighted the fact that embryonic stem cell research represents a social and ethical challenge.
http://en.wikipedia.org/wiki/Stem_cell_controversy
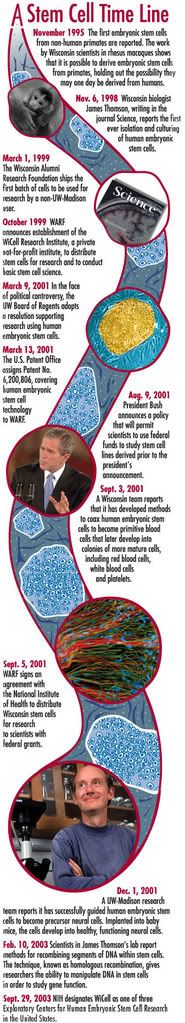
Stem Cell Debate
http://www.cnn.com/SPECIALS/2001/stemcell/

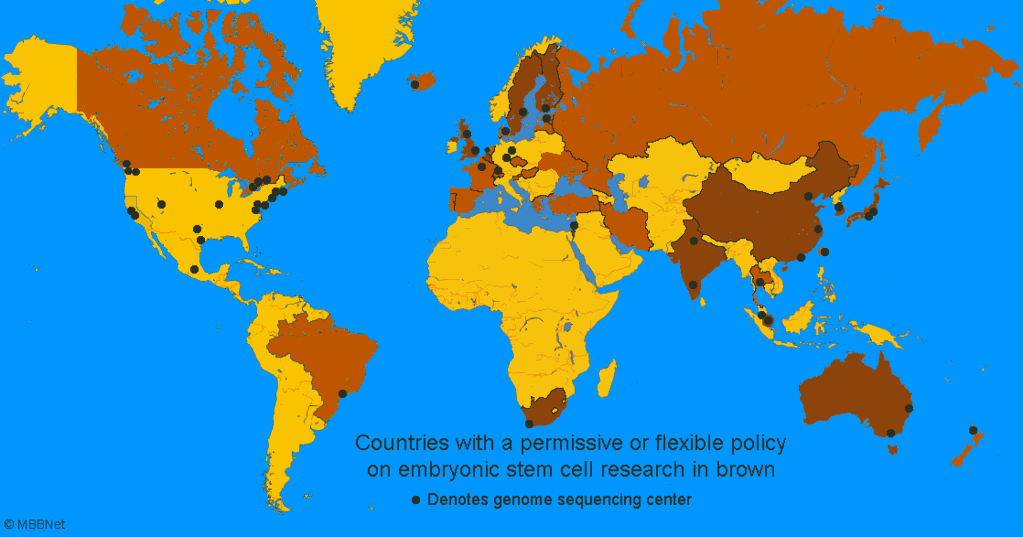
http://mbbnet.umn.edu/scmap.html
Countries colored in brown represent about 3.5 billion people, more than half the world's population. All have a permissive or flexible policy on human embryonic stem cell research and all have banned human reproductive cloning. Population: M = million.
Michael J. Fox (with Parkinson's) talks Stem Cell research
Cyborgs & Stem Cells
Bone Marrow Stem Cells May Cure Eye Disease
http://www.sciencedaily.com/releases/2007/05/070510160859.htm
Science Daily — Adult bone marrow stem cells may help cure certain genetic eye diseases, according to UC researchers.
Scientists have completed a study using mice which showed that bone marrow stem cells can switch roles and produce keratocan, a natural protein involved in the growth of the cornea—the transparent, outer layer of the eyeball. This ability of marrow cells to “differentiate” into keratocan-producing cells might provide a means for treating abnormal corneal cell growth in people.
Winston Whei-Yang Kao, PhD, professor of ophthalmology, and Hongshan Liu, PhD, research scientist in the department of ophthalmology, will present their findings at the annual meeting of the Association for Research in Vision and Ophthalmology being held in Ft. Lauderdale, Fla., May 9 and10.
In the laboratory, the researchers induced corneal abnormalities that mimicked genetic eye mutations and then injected bone marrow stem cells into the corneas to see if they altered the mutations.
The study showed that after only one week, the abnormal corneas of animal models injected with bone marrow stem cells began to change shape and heal.“We found that bone marrow stem cells can contribute to the formation of connective tissues,” Kao said. “If we can change the function of non-corneal bone marrow stem cells by introducing them into human corneas, we can possibly repair the loss of visual sharpness caused by mutations.”
Kao and his coworkers are now planning a clinical trial. If the trial succeeds, Kao said, the procedure could help prevent blindness in future generations who suffer from genetic corneal diseases.
He added that cornea transplants have been successful to some degree but do not always eliminate the problem. ”When the donor cells disappear after a few years, the corneal disease often reoccurs,” he said. “However, if we can place the stem cells inside the cornea, they will repair the lost function of the mutated gene, and stem cells can presumably renew themselves and maintain effective treatment longer, if not forever.”
This study was funded by grants from the National Eye Institute, Research to Prevent Blindness, and Ohio Lions Eye Research Foundation.
Scientists plan stem cell cure for blindness
http://www.reuters.com/article/topNews/idUSL0584115420070605?feedType=RSS
LONDON (Reuters) - British scientists plan to use stem cells to cure a common form of blindness, with the first patients receiving test treatment in five years.
The pioneering project, launched on Tuesday, aims to repair damaged retinas with cells derived from human embryonic stem cells. Its backers say it involves simple surgery that could one day become as routine as cataract operations.
They believe the technique is capable of restoring vision in the vast majority of patients with age-related macular degeneration (AMD), a leading cause of blindness among the elderly that afflicts around 14 million people in Europe.
Some drugs, like Genentech Inc.'s Lucentis, can help the one in 10 patients with so-called "wet" AMD and U.S. biotech firm Advanced Cell Technology is looking at stem cells in other eye conditions. But there is no treatment for the 90 percent with "dry" AMD.
AMD is caused by faulty retinal pigment epithelial (RPE) cells, which form a supporting carpet under the light-sensitive rods and cones in the retina.
The new procedure will generate replacement RPE cells from stem cells in the lab, with surgeons then injecting a small patch of new cells, measuring 4 by 6 millimeters, back into the eye.
U.S. DONOR
The London Project to Cure AMD brings together scientists from University College London (UCL), Moorfields Eye Hospital in London and the University of Sheffield.
It has been made possible by a 4 million pounds ($8 million) donation from an anonymous U.S. donor, who the project's leaders said had become frustrated by U.S. curbs on stem cell work.
Embryonic stem cells are the ultimate master cells of the body, giving rise to all of the tissues and organs. Their use is controversial because many people oppose embryo destruction, although Britain has encouraged such research.
Surgeons at Moorfields have already restored the vision of a few patients using cells harvested from their own eyes, which were moved to a new site. But this process is complicated and only a small number of cells can be moved, limiting its use.
By injecting RPE cells derived from stem cells instead, Dr Lyndon Da Cruz of Moorfields hopes the operation can be reduced to a simple 45-minute procedure under local anesthetic.
"If it hasn't become routine in about 10 years it would mean we haven't succeeded," he told reporters. "It has to be something that's available to large numbers of people."
Similar tests on rats have already proved highly effective.
Pete Coffey of UCL, the director of the project, said he was confident the procedure would work in humans but the team needed to ensure the safety and quality of batches of cells, which would take time.
"The goal is within five years to have a cohort of 10 or 12 patients to put the cells into," he said.
The project, which is non-commercial, was welcomed by patient support groups. Alistair Fielder of the eye research charity Fight for Sight said it represented a real chance to tackle a hitherto untreatable condition.
Stem cell cure for heart attacks
http://www.dailymail.co.uk/pages/live/articles/health/healthmain.html?in_article_id=415156&in_page_id=1774&in_page_id=1774&ct=5&expand=true
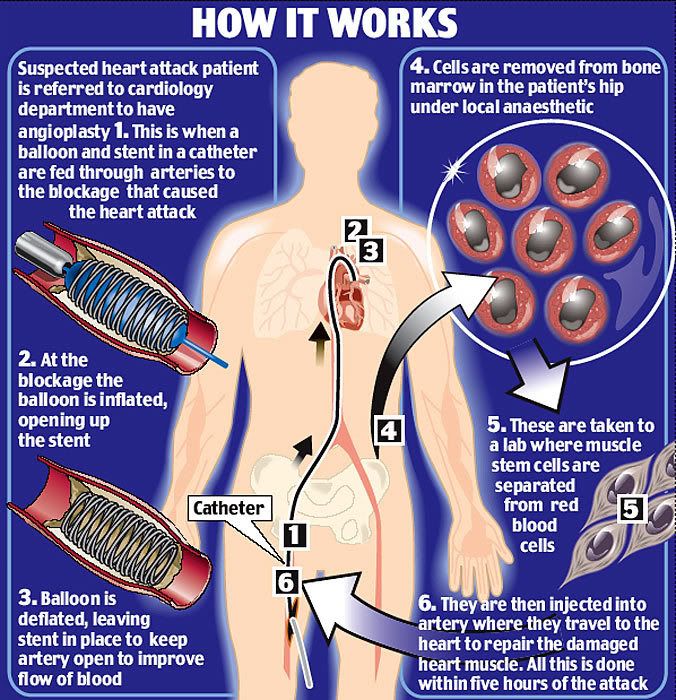
Emergency heart attack patients will be injected with their own stem cells in a dramatic new treatment.
The procedure, being pioneered by British doctors, holds out hope of a 'cure' as the stem cells repair damaged heart muscles.
The low-cost treatment, which involves removing stem cells from the patient's bone marrow, could be given within a few hours of a heart attack.
It is intended to stop patients suffering further attacks and developing heart failure, something existing treatments fail to do in many cases.
If the initial trials in London are successful, the treatment is likely to be extended to NHS hospitals across the country.
Researchers have called the project - the first of its kind in the world - "very exciting" and say it could have a significant impact on the annual toll of deaths from heart disease.
As well as saving lives, it would also reduce the £7billion-a-year burden of heart attacks on the economy through hospital admissions, drug prescriptions and lost working days.
Heart disease is the biggest killer in the UK. Every two minutes someone, somewhere, has a heart attack. When a patient reaches Accident and Emergency, they are currently given a treatment called angioplasty to re-open the blocked artery that triggered the problem.
This boosts survival rates and cuts repeated hospital admissions. But a significant proportion of patients will still be left with damage to their hearts.
Many people can end up with heart failure - in which the heart gradually loses its ability to pump blood and so leaves the person breathless and unable to move around.
Now two London NHS trusts - University College London Hospital and Barts and the London - plan to use stem cells to try to repair the damage immediately after a heart attack.
Stem cells are the basic building block cells that can grow and change into different types of tissue.
They have been shown to help people who already have heart disease by aiding the regrowth of damaged tissue.
The new trials, however, would be the first in the world to give patients the cells within hours of their heart attack in a bid to prevent any heart failure developing at all.
People who suffer a heart attack and are taken to the London Chest Hospital or London Heart Hospital for treatment will be asked for written consent to take part in the study.
After their angioplasty, those who agree will have stem cells removed from their hip using a needle under local anaesthetic.
The stem cells will then be placed in the same artery as the angioplasty - all within five hours of their original attack.
Doctors will then monitor the patients over the following months to establish how effective the stem cell treatment has been at preventing heart failure and repairing the damaged organ.
The research is being led by Professor John Martin, chair in cardiovascular sciences at the British Heart Foundation, and Dr Anthony Mathur of Barts and the London NHS trust.
Professor Martin said last night: "This is the first time in the world that stem cells have been used to stop the damage of acute heart attack.
"It is very exciting. We feel we can make a considerable reducstemtion in deaths and suffering from heart failure."
The cost for each procedure has not yet been worked out. But Professor Martin said it would be very low as the cells come from the patients themselves, it does not prolong their hospital stay and the only expense is on needles and laboratory time separating out the muscle cells.
Dr Mathur said: "If we can demonstrate improvement in the quality of life of patients then this will be a significant step forward in the treatment of heart disease.
"Because the cells are taken from the patient themselves there are minimal ethical issues surrounding this procedure.
"There is also less likelihood of rejection complications."
The research project, which is due to start after Christmas and will involve 100 patients, is being funded by the UK Stem Cell Foundation.
It is a randomised doubleblind controlled study which means half of the patients will be given stem cells and the rest just blood serum, to act as a comparison group.
Chief executive David Macauley said: "This is the first known project of its type in the UK to combine stem cell delivery to the heart with primary angioplasty - where the blocked arteries of heart attack patients are opened as quickly as possible."
The doctors do not yet know whether the injected stem cells will turn into heart tissue and repair the damaged areas.
This is because they are using adult stem cells from the patient's own bone marrow, which are not always very adaptable.
However, trials in other countries on heart patients - though not immediately after an attack - showed that their own stem cells successfully grew into heart tissue. Most medical research uses stem cells from human embryos because these are more adaptable and can grow into any type of bodily material.
A heart attack is caused by a blockage which prevents blood reaching the muscle and usually leads to a crushing pain in the chest, shortness of breath and dizziness.
Most people who suffer a heart attack remain conscious.
However many will go on to suffer a cardiac arrest, which is when the heart stops and the person loses consciousness and stops breathing.
Anyone who was unconscious at the hospital would not be able to take part in the trial as they could not give their consent.
Professor Peter Weissberg, medical director of the British Heart Foundation, said: 'In the future, stem cells could herald a new frontier for heart patients.
"We welcome the research that helps us understand the potential role stem cells may yet play in treating heart disease.
"We hope that this study will compliment the BHF's existing research in this area."
Brain stem cells to cure diabetes
Transplants could ultimately remove the need for daily insulin injections
US scientists believe they could use brain stem cells to cure diabetes.
http://news.bbc.co.uk/1/hi/health/4480875.stm
Although the work is not yet ready to be tested on human patients, results in animals have been promising, say the Stanford University researchers.
They were able to coax the immature brain cells to develop into the insulin-producing islet cells that are lacking in diabetes.
Eventually, these could be used for curative transplants, the scientists told the journal PLoS Medicine.
Stem cells
Scientists have already been looking at using stem cells taken from embryos to treat diabetes.
These are primitive "master" cells that can be programmed to become many kinds of tissue.
However, there have been concerns that these cells can turn cancerous, are difficult to work with in the laboratory.
Dr Seung Kim and colleagues looked at whether stem cells taken from the brain might work just as well and avoid some of these issues.
Dr Kim said: "When you look at islet cells you realise that they resemble neurons."
In some insects, such as fruit flies, the cells that produce insulin and regulate blood sugar are also neurons.
Chemical cocktail
Dr Kim's team found that when they added a cocktail of chemicals to brain stem cells, taken from aborted fetuses, the cells changed and, although they were not identical to islet cells, they were able to produce insulin in response to blood sugar levels.
To find out whether these cells would work, they transplanted them into a cavity in the kidney in mice where other types of insulin-producing cells have been found to survive before.
When the blood sugar went up in these mice, the transplanted "mature" brain stem cells again released insulin.
Four weeks later, the cells were still alive and producing insulin and none had turned cancerous.
Dr Kim said, although it was early days, the work suggested that stem cells could be used to replace islet cells and free people with type 1 diabetes daily insulin injections.
Some patients have already received transplants using islet cells taken from living relatives or dead donors.
Dr Angela Wilson, director of research at Diabetes UK said: "This is an interesting result and may provide another avenue to explore in our search for a cure for diabetes.
"However, the work is in the very early stages of development and has yet to be reproduced in humans.
"We'll certainly be following the progress of this research with interest."
Facts on Stem Cells
http://www.washingtonpost.com/wp-dyn/articles/A25071-2004Aug22.html
Key Facts About Stem Cell Research
http://www.curesforcalifornia.com/page.php?id=4
Stem cell research is perhaps the most exciting medical technology of the 21st Century. Stem cells hold the promise of treatments and cures for more than 70 major diseases and conditions that affect millions of people, including diabetes, Parkinson’s, Alzheimer’s, cancer, multiple sclerosis, Lou Gehrig’s Disease (ALS), spinal cord injuries, blindness, and HIV/AIDS.
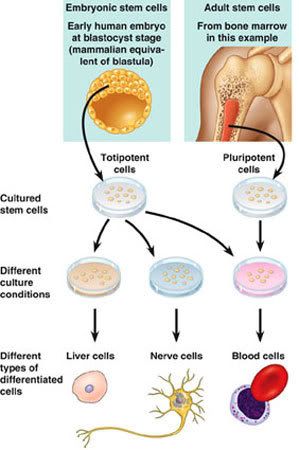
Stem Cells are the Body’s Basic Building Blocks
Stem cells have two unique qualities: they can copy themselves for an unlimited period of time and they can grow into all of the body’s different cell types.
When a stem cell copies itself, each of the two new cells can either remain a stem cell or become another type of cell such as a muscle cell, a blood cell, or a brain cell.
Some stem cells grow into organs, bones and tissues of the body, but other stem cells work in its immune and repair systems. Called “adult” stem cells, they create replacement cells for those that are lost because of injury, disease or the normal wear and tear of aging.
Adult stem cells can be used for research and treatment, but most researchers believe that “embryonic” stem cells have much more potential to study, treat and cure diseases.
Not all stem cells are alike.
Adult stem cells are called “multipotent.” This means they can turn into several different kinds of cells within the same basic cell type. For example, adult stem cells found in bone marrow can turn into all of the different types of cells found in blood. Research on adult stem cells has been funded for over 40 years and many successful therapies have been developed.
Embryonic stem cells are called “pluripotent.” This means they can turn into all of the body’s cell types except egg and sperm cells. Embryonic stem cells were first discovered in 1998 and research on them has not yet been well funded. Scientists believe that the potential of embryonic stem cells is even greater than adult stem cells.
Embryonic stem cells are blank, unspecialized cells found in the interior of a blastocyst. A blastocyst is a mass of 150 or so cells that is smaller than a period at the end of this sentence. It develops approximately 5-14 days after an egg is fertilized.
Scientists have been able to get stem cells to grow into complex cells with special functions, like the cells of the pancreas that produce insulin or heart muscle cells.
A research process called somatic cell nuclear transfer (SCNT), sometimes referred to as “therapeutic cloning”, allows scientists to produce embryonic stem cells without using sperm to fertilize an egg. In this process, there is no implantation in a uterus and therefore no pregnancy.
Using new tools like somatic cell nuclear transfer (SCNT), scientists hope to make cell lines that are specific to a patient and/or a disease. This will help them study diseases in a powerful new way and help them discover better treatments and cures.
Stem Cell Research is Developing Breakthrough Cures & Offering New Hope
Life-saving stem cell treatments are already successful and in widespread use for some diseases. For example, bone marrow transplants use stem cells to treat leukemia.
Stem cell research could lead to treatments that save millions of lives and improve the quality-of-life for millions more.
Scientists are working on a wide range of stem cell-based therapies. They have created “tissue patches” for burn victims. They have grown islet cells that produce insulin to treat diabetes and healthy brain cells to ease the symptoms of disorders like Parkinson’s disease. They have genetically changed cells and then used them to deliver healing or protective agents to injured or diseased areas of the body.
Broad Consensus Exists in Support of Stem Cell Research
Stem cell research has strong support by many notable groups including the American Medical Association and the National Health Council.
63% of Americans support embryonic stem cell research.
55% of Texans favor medical research using embryonic stem cells.
70% of Kansas voters want to allow SCNT research in Kansas.
63% of Missouri voters approve of embryonic stem cell research.
64% of Americans support federal funding of research on chronic diseases using stem cells taken from human embryos.
Stem cell cure hope closer
http://www.theage.com.au/articles/2004/06/25/1088046261808.html?from=storylhs
Cures for diabetes, Alzheimer's and spinal cord damage could be as little as five years away, after the landmark creation of more than one million stem cells from a single Australian embryo.
The work of Sydney IVF's research laboratories, the achievement is the first time Australian scientists have cultured embryonic stem cells from an Australian embryo on home turf.
Although legal hurdles have set the nation's stem cell research two years behind the United States-led cutting edge, Sydney IVF medical director Professor Robert Jansen is confident of catching up.
"There are several hurdles, ... but there's a small chance it will be inside five years," Prof Jansen said of the possible therapeutic uses of cultured embryonic stem cells.
One of those hurdles has already been overcome by research director Dr Tomas Stojanov and his team - the need to grow the cells on human "feeder layers" rather than culturing them on mouse cells.
However, safety and anti-contamination guidelines are yet to be drawn up, the issue of tissue rejection by stem cell recipients must be overcome and scientists need to work out how to generate enough cells to meet demand.
At the moment ... there are not enough lines for research and future clinical use," Dr Stojanov said.
The Sydney team is now trying to automate the process of extracting viable cells from unwanted five-day-old embryos, so they can produce stem cells in much greater volumes.
Millions of cells would need to be injected as a treatment for just one patient, Dr Stojanov explained.
The cells cultivated by the Sydney IVF team remain undifferentiated, meaning they have the potential to turn into any cell in the human body.
There's already a huge demand for the IVF clinic's stem cells, from medical research scientists who plan to turn them into nerve, heart and pancreatic cells, among others.
"The obvious direction of the research is the treatment of serious childhood and adult diseases where cells are lost or damaged or destroyed and can't be replaced by the body's own stem cells," he said.
"That's true of some tissues more than others, particularly the brain and spinal cord (and) in childhood diabetes, where the insulin-producing cells of the pancreas have lost their function or are so depleted in number they can't do their job."
Conditions that have the potential to be treated with stem cells include Alzheimer's, Parkinson's disease, multiple sclerosis, motor neurone disease, spinal cord and brain damage, diabetes and heart conditions.
The clinic won licences in April to cultivate stem cells from as many as 600 embryos, after the federal government legislated to allow research on human embryos.
In May this year the clinic thawed an embryo frozen in March 2000, then extracted cells from its centre and placed them on a human cell-based "feeder" culture to grow.
There are strict criteria for the harvesting of stem cells - the embryos must have been created for the purpose of assisted reproduction and the permission of its parents must be obtained.
The embryo also must be one that would otherwise have been discarded, but that doesn't excuse the research in the eyes of Right To Life Australia or the Catholic church.
"Two wrongs don't make a right - they should not have created excess embryos in the first place," said Margaret Tighe, president of Right To Life Australia.
"Quite clearly this involves the destruction of a very small human being for the benefit of other human beings."
Even if embryonic stem cells could save a loved one, Ms Tighe said she would remain against the research going ahead.
The Life Office, an agency of the Catholic Archdiocese of Sydney, condemned the research as unethical and scientifically unnecessary.
"Although embryonic stem cells have been used for research around the world for some time, there have been no human trials and there are still no approved medical treatments," Life Office executive officer Dr Brigid Vout said.
"It is research using stem cells from adults which holds the greatest therapeutic promise.
"Adult stem cells are already being successfully used to cure disease or overcome injury in patients."
Prof Jansen acknowledged that a cure is yet to be effected using embryonic stem cells, but pointed out that the research had only been given legislative approval after vigorous and protracted debate.
"I'm very proud that Australia has risen to the occasion legislatively to permit this," he said.
Prime Minister John Howard also reiterated that the work was legal.
"I support very much the law that was agreed upon at the premiers' conference in 2002 and I want that law observed," Mr Howard told reporters.
More than half of the clients at Sydney IVF were in favour of donating their excess embryos for stem cell research, Prof Jansen added.
"I think most couples, when they've had such an amount of difficulty getting pregnant, realise that their success has depended on people who've donated embryos before for research," he explained.
The couple who donated the embryo used to cultivate the stem cells revealed this week were "absolutely delighted", Prof Jansen said.
"They expressly asked that they be informed when the embryos they declared to be excess were entered into a scientific study," he said.
"When I phoned them to say they'd produced the first stem cell line they were over the moon."
Cord Blood Stem Cell Transfusion Saves 3-Year-Old Child
Stem Cells in Baby Teeth!
Reeve's legacy:
stem cell research
http://www.msnbc.msn.com/id/6224513/
The legacy of actor Christopher Reeve won't be for soaring through the air and leaping tall buildings as Superman, but for bringing public attention to spinal cord research while in a wheelchair, medical experts say.
Christopher Reeve's message to United Nations'













![Brotherhood" (2006) [TV-Series]](http://photos1.blogger.com/x/blogger2/1421/379621144723082/211/z/425926/gse_multipart33129.jpg)







No comments:
Post a Comment